My son has a Statistical Physics for Babies book. It came as a set with Astrophysics for Babies, Optical Physics for Babies and the ABC’s of Engineering ( “E is for Electricity!”). I was reading him Statistical Physics for Babies the other day and I learned something new. I learned why my house never stays clean.
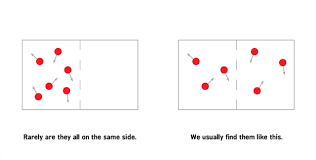
The author describes “entropy” to babies as the number of different combinations in a system. Low entropy being few or one combination, and high entropy being a lot of different combinations. The book uses colored balls organized in a square space to illustrate this point. Organizing all twelve balls into a perfect equal grid is low entropy. There is only one combination in which this happens. High entropy is when the balls are everywhere, because statistically speaking, there are a greater number of combinations of the balls flying everywhere. The last two pages read, “Things naturally go from organized to chaos. To go from high entropy to low entropy you must add effort and energy” Bingo!

This is why our house never stays clean.
It’s the second law of thermodynamics. Entropy is always increasing. Of course!
When the home is clean, everything is in its place. That’s low entropy. There is only one combination. As the day or week goes on things get moved, clothes on the floor, dishes pile up, etc. Things naturally go from organized to chaos. That’s physics! So mess is inevitable. It would be an impossible expectation to try to keep everything clean all of the time. As we have learned from thermodynamics, in order to keep everything clean (low entropy), you would need to exert effort and energy alllll of the time. Which is unrealistic and unhealthy.
So next time your house is a mess, just say, “that’s entropy!”.
By the way, I highly recommend all the physics for babies books by Chris Ferrie. They are such a great way to introduce kids to math and science concepts in a fun and engaging way. They lay the foundation for future learning and encourage them to ask questions. Andddd you might learn something too!